MEDICAL PRODUCTS
.jpg)

Asfora “SAMBA™ Screw” System Instrument Kit
The SAMBA™ Screw” System required the development of a set of custom designed instruments (tools) which are presented to the Surgeon in the operating room to implant the SAMBA™ Screws. The instruments in the kit include a Drill Guide, Pin Sleeve, Striker Tube, Drill Gauge, Adjustable Pilot Hole Drill Bit, Driver Bit, Parallel Spacer Mechanism and a Bone Substitute Packing Plunger Assembly. All instruments are secured in a custom designed Sterilization Tray which meets the requirements for autoclave sterilization.
The Asfora “SAMBA™
Screw” System Instrument Kit project scope included conceptual
design, product testing, engineering drawings and technical support
through release for manufacture.

Asfora “SAMBA™ Screw” System
Working with Dr. Wilson Asfora of Medical Designs, LLC, SMP designed a proprietary, Class 2, implantable bone fixation screw system used to fuse the Sacroiliac Joint to alleviate joint disruptions.
 The
project scope included conceptual design, FEA
analysis, product testing, engineering drawings
and technical support through release for manufacture.
Our project activities also included providing
regulatory support to Medical Designs for Risk
Management, Design Verification and Validation,
FEA Reports and other deliverables required for
the FDA 510(k) regulatory submission.
The
project scope included conceptual design, FEA
analysis, product testing, engineering drawings
and technical support through release for manufacture.
Our project activities also included providing
regulatory support to Medical Designs for Risk
Management, Design Verification and Validation,
FEA Reports and other deliverables required for
the FDA 510(k) regulatory submission.
The SAMBA™ Screw is designed to be pre/post packed with bone growth substitute to naturally stimulate bone growth after surgical implantation. There are five SAMBA™ Screw lengths ranging from 25mm to 50mm in 5mm increments. The SAMBA™ Screws are implanted with a minimally invasive surgery protocol developed by Dr. Wilson Asfora.
SMP Tech, Inc. completed the industrial design and packaging of the PEA POD® Infant Body Composition System for Life Measurements, Inc. The enclosure system is composed of a combination of sheet metal and pressure formed parts. The Peapod provides vital information for optimal infant growth and nutrition for infants. The system uses air displacement plethysmography to assess body composition in infants. The accurate assessment of infant body fat and fat free mass provides important data to assist in addressing several infant situations, including optimizing individual nutritional and pharmacological requirements.

Implantable Medical Device
Anterior Cervical Plate System
SMP designed a proprietary Anterior Cervical Plate System (AACP) for Medical Designs, LLC based on input from Dr. Wilson Asfora. The Cervical Plate is a Class 2 spinal intervertebral body fixation orthosis. The project scope included conceptual design, FEA analysis, product testing, engineering drawings and technical support through release for manufacture. Our project activities also included providing regulatory support to Medical Designs for Risk Management, Design Verification and Validation, FEA Reports and other deliverables required for the FDA 510(k) regulatory submission. The AACP system met or exceeded all predicate devices in performance.
The Anterior Cervical Plate System is used to provide temporary stabilization of the anterior spine during the development of cervical spine fusions and is comprised of a dual-contoured Cervical Plate and constrained Locking Screws. There are four Levels of Cervical Plates (five lengths per Level) which provide different Plate lengths and mounting hole spacing to fit a wide variety of patients. Each Locking Screw is captured in the Cervical Plate mounting holes by proprietary “Gripper” tabs which prevent the Bone Screws from coming out once they are surgically installed.

Cervical Plate Bender
SMP designed a proprietary Anterior Cervical Plate Instrument used to bend a titanium cervical plate. The project scope was to develop a custom surgical instrument to be used to alter the contour of the Medical Design’s proprietary Anterior Cervical Plate in the operating room.
The Bending Instrument was required to provide the surgeon with a simple, yet reliable means of altering the curvature of the Cervical Plates without affecting the restraining features located adjacent to the screw holes. This Bending Instrument was also required to fit into a custom designed Sterilization Tray which is presented to the Surgeon in the operating room and is also used during the autoclave sterilization process.
The scope included conceptual design, product testing, engineering drawings and technical support through release for manufacture. Our project activities also included providing regulatory support to Medical Designs for Risk Management, Design Verification and Validation, FEA Reports and other deliverables required for the FDA 510(k) regulatory submission.

Collimator System
This system was developed for use with the Varian Medical Clinac Linear Accelerator to treat Macular Degeneration, the age-related loss of vision. The collimator system for the Macular Irradiator System includes an Optics/Collimator that is attached to the Varian's Clinac Linear Accelerator. The Collimator assembly provides a small elliptical beam for radiation treatment of the eye. The system includes two video cameras to provide precision alignment to the patient's eye.

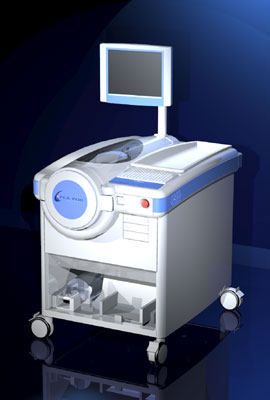
The system shown was developed for use with the Varian Medical Clinac Linear Accelerator to treat Macular Degeneration the age-related loss of vision. The computer system includes the vision system and safety interlock system. It is packaged on a Mobil cart to expedite its use inside or outside the treatment room.